The Importance of Knowing the Ct Value at which a SARS-CoV-2 PCR Test is Positive
Throughout the COVID-19 pandemic the SARS-CoV-2 PCR test (often referred to as the COVID PCR test) has played the major role in diagnosis of individual patients and collection of epidemiological data.
To date, this test has been reported as being either positive or negative, with no indication on the report as to whether the result was strongly positive or only weakly positive. This has made it difficult for people and their physicians to know whether, at one extreme, a person is infected with a huge load of active “live” contagious virus that greatly threatens the person and those around them; or, at the other extreme, the test has detected only a trace amount of inert, non-infectious, “dead” viral debris that poses no threat to the person or to others and might even represent a false positive result.
Not knowing where a person fits along the above spectrum hampers optimal care of patients with severe life-threatening COVID illness, creates unnecessary fears regarding people at the other end of the spectrum, and adversely affects the quality of state and national epidemiological data.
The Ct value at which a person’s SARS-CoV-2 PCR test is positive can shed considerable light on the significance of a positive test. Below is an explanation of the importance of knowing the Ct value at which a SARS-CoV-2 PCR test becomes positive:
Ct = Cycle threshold; Ct = the number of amplification cycles needed before the test is able to detect presence of viral material in a specimen. The higher the number of amplification cycles needed before the test is able to detect viral material (i.e., the higher the Ct number), the lower the viral load and the less sick and contagious the person is likely to be.
If a test becomes positive after only 12 amplification cycles (i.e., becomes positive at a Ct of 12), the viral load is very high—at least 100,000,000 copies per microliter. [1-3] If the test becomes positive after 22 cycles (at a Ct of 22), the viral load is approximately 2,500,000 copies/mL. [4-5] If the test becomes positive only after 37, 40, or 45 amplification cycles, the result most likely represents either a false positive, or is a true positive due to only a trace amount (less than 100 copies, even just a few copies) of inert, non-contagious, “dead” SARS-CoV-2 viral debris (assuming the test is truly capable of always accurately identifying such a tiny amount of viral debris). [2, 6, 7]. Rarely, a positive test at a high Ct is identifying an asymptomatic/pre-symptomatic person who has very recently become infected and might soon have a high viral load (low Ct), but this possibility can be evaluated by carefully following the person and repeating the test within 3-4 days, to see if symptoms develop and/or the Ct drops.
The PCR test for the SARS-CoV-2 virus is a good test when it is properly manufactured, properly conducted, used in an appropriate setting (e.g., in the evaluation of inpatients with COVID-like clinical features), and properly interpreted by carefully and fully taking Ct values into account. (This statement assumes that the manufacturers of the tests have been truthful in their claims that their tests are specific for the SARS-CoV-2 virus—i.e., they detect markers that are unique to the SARS-CoV-2 virus and not found on any other viruses; i.e., there is no cross-reactivity.)
It is not a reliable test, however, when used in the screening of asymptomatic or only mildly or non-specifically symptomatic individuals, if the test is positive only after 33 or more cycles of amplification and this full information is not reported to patients and their physicians.
Unfortunately, it is very difficult to know with certainty whether a positive result at a Ct of 33 or higher represents a false positive or an accurately identified trace amount of SARS-CoV-2 viral material. The test was not designed to be reliably accurate after so many amplification cycles. When the test is used in an appropriate setting and the test is positive at a Ct of 30 or less, the false positivity rate is probably less than 4% (perhaps only 1-2%, as the test manufacturers claim). However, when the test is used in a surveillance setting and is “positive” at a Ct of 33 or higher (particularly at 37 or higher) in a person who is asymptomatic or has had only mild or non-specific symptoms, the exact false positivity rate is currently unknown and could be quite high—possibly as high as 70%. [6]
Based on what is currently scientifically known, it is best (most accurate) to label any test result that is “positive” at a Ct higher than 32 as an “inadequately interpretable” result. It is not scientifically sound and, in fact, is misleading and harmful, to label people with a positive test at a Ct of 33 or higher as a “new COVID-19 case,” especially if they have been asymptomatic or have had only mild or non-specific symptoms. More accurately, they are people with an “inadequately interpretable” result who, furthermore, are unlikely to be infectious [2, 8]. Regarding this latter point, please see Graph 1 (after the References section), which points out that it is extremely unlikely that a person with a positive test at a Ct of 35 or higher is infectious.
For the above reasons, experienced PCR scientists recommend stopping the PCR test after 30 (or 32 at the most) amplification cycles, because positive results obtained after 32 or more cycles are unreliable (inadequately interpretable) [2] and are not associated with contagiousness [2, 8].
Unfortunately, to date, SARS-CoV-2 PCR tests are still being reported only as positive or negative, with no indication of how strongly or weakly positive. Although Ct results have always been available for each individual test since the beginning of the pandemic (with rare exception), Ct results have not been routinely reported or used for clinical or epidemiological purposes. This has been the case throughout the USA and most of the world.
It has also been unfortunate that most SARS-CoV-2 PCR tests are set to perform 40, 45, even 50 amplification cycles in their effort to detect viral material. (This varies from one test kit to another—see Table 1 after References.) That is, if a person’s specimen is negative after 30 amplification cycles, further cycles are, nevertheless, performed (up to 50 cycles with some tests), looking for evidence of tiny amounts of viral material. Only if no viral material is detected after 40, 45, or 50 cycles (whichever number the test system sets as the stop point) is the test declared negative. Even if a test becomes positive only after 45 or 50 amplification cycles, it is declared a positive test (without any mention of the Ct value) and the person tested is declared a “new COVID case.” This practice is not scientifically sound and is misleading.
When a person is told they have a positive SARS-CoV-2 PCR test, they deserve (morally, ethically, and practically) to know how strongly positive their test is and what their result means. Does their result mean they are carrying a huge viral load, are very contagious, and should be very worried about themselves and those with whom they have been in contact? Or are they carrying only a tiny amount of dead, non-contagious viral debris that represents no threat to them or others? Or are they in an asymptomatic/pre-symptomatic phase, with a low viral load that could soon accelerate? Or does their result represent a false positive?
Knowledge of the Ct value at which a person’s test is positive can help greatly to answer the above critically important questions. But, again, to date, Ct values of positive tests have not been made available to patients, physicians, public servants, or the public.
Having emphasized the importance of knowing the Ct value at which a test is positive, it is important to also emphasize that there are limitations to the information provided by the Ct value. The Ct value is not a true quantitative test of viral load; it just provides a rough and indirect (but, nevertheless, very helpful) estimate (a good, educated guess) of what the viral load might be. It is true, too, that if the same specimen is tested with 3 different SARS-CoV-2 PCR tests, each might be positive at a different Ct value (e.g., at a Ct of 16 in one test, 20 in another test, and 22 in the third—but not at 37 or 45 in one of the three). For these reasons Ct values need to be interpreted with caution and in clinical context, particularly until more data on Ct values of positive tests have been collected and fully analyzed.
In the meantime, it is far better to have a SARS-CoV-2 PCR test report that includes the Ct value at which the test was positive, than to have a report that only says positive (or negative) without any Ct information provided. Though imperfect, the estimate of viral load and risk of contagiousness offered by the Ct value is far more valuable than no estimate at all, especially if the Ct value is carefully interpreted and placed into clinical context.
When in early November 2020 the CDC reported that 100,000 “new COVID cases” (meaning, more honestly, new instances of a person having a “positive” SARS-CoV-2 PCR test result) were occurring per day in the USA, neither the individuals with the positive tests, their physicians, their public health departments, the CDC, the NIH, WHO, Johns Hopkins University, or the public knew what percentage of those 100,000 tests were positive at a Ct >32 and what percentage were positive at a Ct of 30 or lower—because, to date, the Ct values at which tests have been positive have not been reported or taken into consideration.
It would be enormously beneficial if we, as a nation, were to report, study, clinically use, learn from, and base public dialogue and public policy (at least in part) on the Ct values of positive tests. This would include retrospective (since the beginning of the pandemic) and prospective reporting of the Ct values of all positive tests. By doing so, we would do the American people and the rest of the world a great favor.
Medically, morally, ethically, and practically— individuals with positive PCR tests, as well as physicians, epidemiologists, public policy makers, and the public— deserve to know, and need to know, the Ct value at which a SARS-CoV-2 PCR test is positive. Without Ct information, interpretation of the number of “new COVID cases,” “new COVID hospitalizations” and “new COVID deaths” is severely compromised, as is public policy and the care of individual patients.
From now on, when a person is told that their SARS-CoV-2 PCR test is positive, they and their physicians would be wise to ask, “At what Ct value was the test positive?” And when the public is told that 100,000 new COVID cases have been occurring per day, the public and their public servants would be wise to ask, “What percentage of those 100,000 were positive at a Ct of 33 or higher (particularly a Ct of 37 or higher)?”
Such questions and their honest answers would create more accurate and useful clinical and epidemiological data, facilitate more healthy public dialogue, and stimulate much-needed critical thinking—all of which are essential for successful resolution of the COVID-19 pandemic. True science and true democracy depend on high-quality data; critical thinking; and healthy, accurately informed, public dialogue.
For further, more detailed discussion of Ct values, including caveats about Ct information, please see the original full article, “The Importance of Knowing the Ct Value at which COVID PCR Tests are Positive,” which may be found on the “Notes from the Social Clinic” website: https://notesfromthesocialclinic.org/the-importance-of-knowing-the-ct-value-at-which-covid-pcr-tests-are-positive/
Robert Rennebohm, MD
Email: rmrennebohm@gmail.com
Website: https://notesfromthesocialclinic.org/
About the Author: https://notesfromthesocialclinic.org/about-me/
REFERENCES:
- Tom MR, Mina MJ. To Interpret the SARS-CoV-2 Test, Consider the Cycle Threshold Value. Clin Infect Dis. 2020 May 21: ciaa619. Published online 2020 May 21. doi: 10.1093/cid/ciaa619
- TWiV 640: Test often, fast turnaround, with Michael Mina. https://youtu.be/kDj4Zyq3yOA
- Your Coronavirus Test is Positive. Maybe it shouldn’t be. Interview with Michael Mina, MD. Published August 29, 2020; Updated September 17, 2020. https://www.nytimes.com/2020/08/29/health/coronavirus-testing.html
- Bryan A, Fink SL, Gattuso MA, et al., SARS-CoV-2 viral load on admission is associated with 30-day mortality. Open Forum Infect Dis. 2020 Dec; 7(12): ofaa535. Published online 2020 Nov 3. doi: 10.1093/ofid/ofaa535
- Perchetti GA, Nalla AK, Huang ML, et al. Validation of SARS-CoV-2 detection across multiple specimen types. J Clin Virol. 2020; 128:104438. doi: 10.1016/j.jcv.2020.104438
- Francesca F, et al. Detection of SARS-COV N2 Gene: Very low amounts of viral RNA or false positive? J Clin Virol. 2020 Dec; 133: 104660. Published online 2020 Oct 14. https://doi.org/10.1016/j.jcv.2020.104660
- Katz AP, et al. False positive reverse transcriptase polymerase chain reaction screening for SARS-CoV-2 in the setting of urgent head and neck surgery and otolaryngologic emergencies during the pandemic: Clinical implications, Head Neck 42 (7) (2020) 1621–1628, https://doi.org/10.1002/hed.26317
- Jaafar R, Aherfi S, Wurtz N, et al. Correlation Between 3790 Quantitative Polymerase Chain Reaction–Positives Samples and Positive Cell Cultures, Including 1941 Severe Acute Respiratory
Syndrome Coronavirus 2 Isolates, Clinical Infectious Diseases, ciaa1491, https://doi.org/10.1093/cid/ciaa1491
See Graph and Table below:
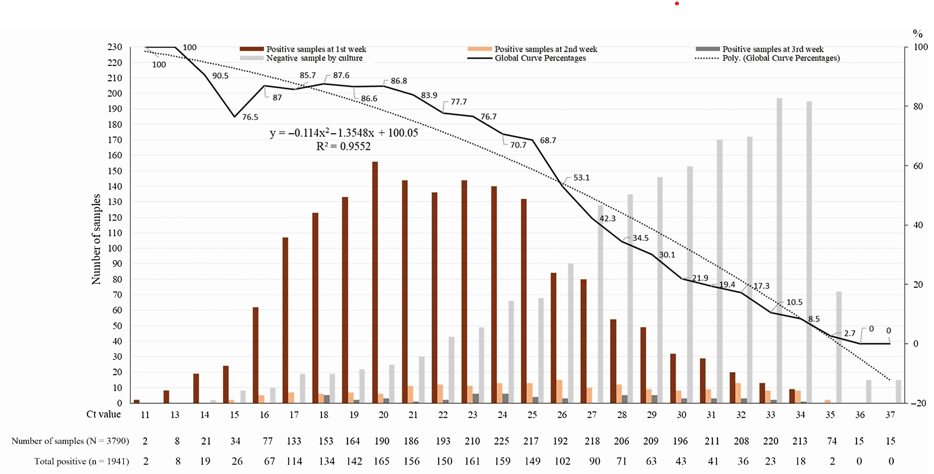
GRAPH 1: Percentage of positive viral culture of SARS-CoV-2 PCR positive naso-pharyngeal samples from COVID-19 patients. No sample that was positive at a Ct >35 had a positive culture. (Reference 18: Jaafar R, Aherfi S, Wurtz N, et al. Correlation Between 3790 Quantitative Polymerase Chain Reaction–Positives Samples and Positive Cell Cultures, Including 1941 Severe Acute Respiratory).
TABLE 1:
The number of amplification cycles that various commercial SARS-CoV-2 PCR Tests are set to perform in their effort to detect viral material:
- Gnomegen: 39 cycles
- GK: 40 cycles
- In Bios-Aires: 45 cycles
- Xpert Xpress: 45 cycles
- Luminex: 45 cycles
- Quest: 50 cycles
0 Comments